Reconstruction of massive full‐thickness abdominal wall defect: successful treatment with nonabsorbable mesh, negative pressure wound therapy, and split‐skin grafting
Clin Case Rep. 2016 Oct; 4(10): 982–985.Published online 2016 Sep 7. doi: 10.1002/ccr3.649PMCID: PMC5054475PMID: 27761251 Dogu Aydin, 1 Ida Felbo Paulsen, 1 Vibeke Egerup Bentzen, 1 Sami Asadzadeh, 2 and Lisbet Rosenkrantz Hölmich
 1
1
Case Report
In May 2014, a 51‐year‐old morbidly obese woman (BMI 48), suffering from non‐insulin‐dependent diabetes, presented at the emergency department with a 20‐year‐old incisional hernia that had become incarcerated. At admittance, the abdomen was described with horizontal and vertical scars from former surgery and a discolored midline hernia measuring 30 × 50 cm. Acute CT of the abdomen demonstrated small bowel obstruction, and an immediate explorative laparotomy was performed via the vertical scar. Perioperatively, the abdominal wall defect measured 15 × 10 cm, and the ventricle, colon, and small intestine were encapsulated in a subcutaneous cavity. The lower 150 cm of the small intestine was cyanotic and torqued around the mesentery; detorquing resulted in revitalization. The intra‐abdominal cavity was found shrunken and too small to contain the intestinal organs. Consequently, the fascia was split proximally and distally of the hernia port; however, the abdominal wall could not be closed without the use of three pieces of nonabsorbable mesh each measuring 35 × 35 cm (Fig. (Fig.1).1). The intra‐abdominal organs were enclosed in the new cavity, and the skin was closed above the cavity.
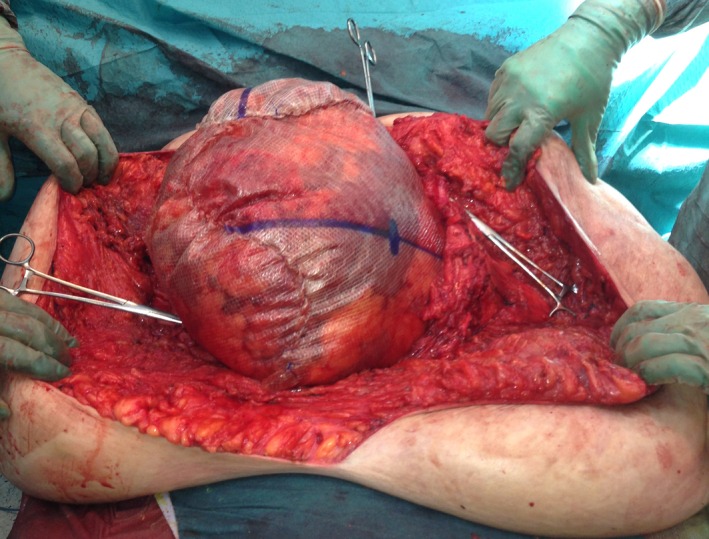
Open in a separate windowFigure 1
Perioperative photograph with three pieces of synthetic mesh sewn together and to the facial edges (shown with peans) to substitute the abdominal wall.
In the following days, the patient gradually developed necrosis of the skin covering the mesh. The wound was revised and necrotized tissue excised leaving a 40 × 50 cm defect with the underlying mesh being intact. Thereafter, the patient was treated with NPWT and surgically revised or cleansed and dressed every other or third day for a period of 8 months, aiming for granulation tissue formation upon the mesh. General anesthesia was used for the first 3 months, and thereafter, wound care was provided with the use of intravenous sedatives and pain medication, but no anesthesia. After another 2 months, wound dressings were performed in the ward without medication, and 5.5 months after the hernia operation, the patient was discharged and wound dressings were performed in the outpatient clinic. One specific nurse was dedicated to the task. After a total of 8 months, the mesh was covered with granulation tissue without infection (Fig. (Fig.2)2) and split‐skin grafts were harvested from both thighs and transplanted to the defect. The patient afterwards developed a low‐output fistula from the small intestine, treated conservatively (Fig. (Fig.3).3). The fistula sealed 6 months postoperatively. After immediate take of the transplanted skin, the mesh started to erode the overlying vital transplant and this problem worsened as the patient was mobilized (Fig. (Fig.4).4). The mesh was very stiff, and wrinkles damaged the overlying skin. A long period of careful and meticulous removal of the exposed mesh was performed bedside or later in the outpatient clinic with tiny bits or threads being removed successively. Now, two years after, a thread can occasionally be removed, but more and more seldom.
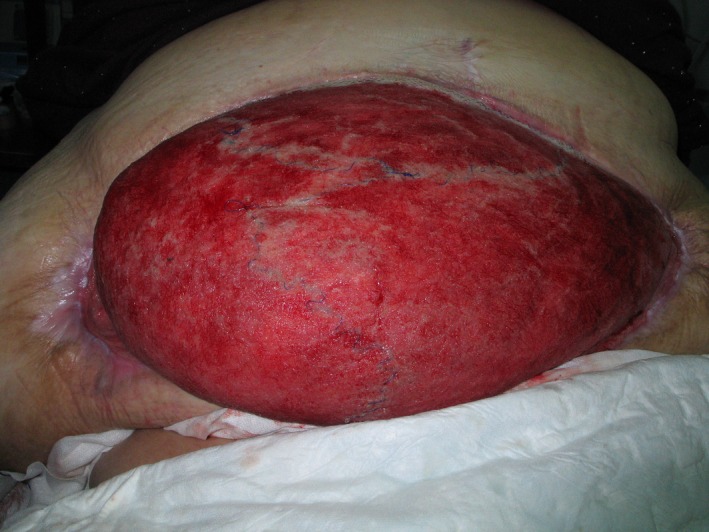
Open in a separate windowFigure 2
After eight months of VAC treatment and just prior to split‐skin transplantation. Note the mesh is almost invisible and the abdominal skin has reattached to the lateral aspects of the truncus.
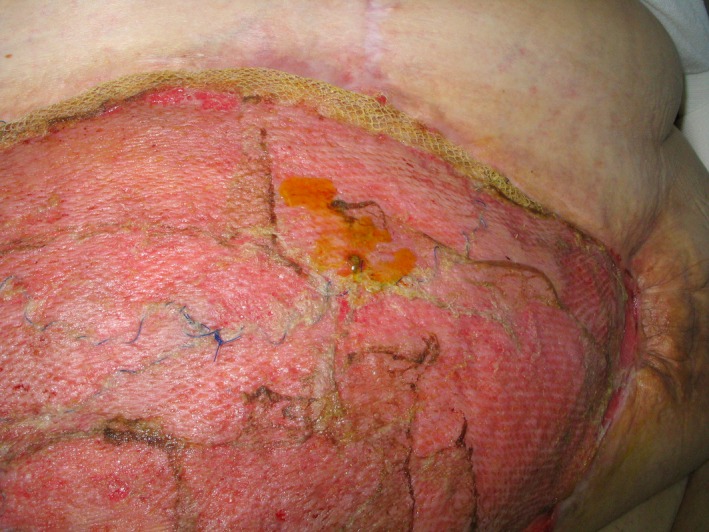
Open in a separate windowFigure 3
One week after split‐skin transplantation. Note good graft take, folding of the mesh in the upper aspect of the defect and yellow secretion from what turned out to be a high small bowel fistula with low output.
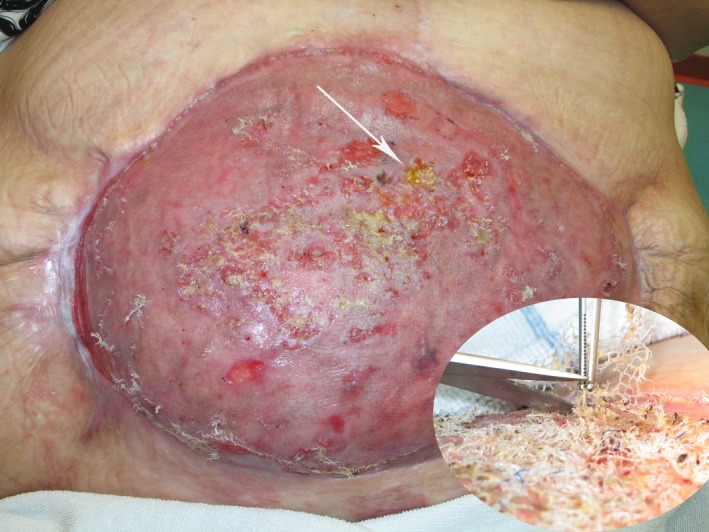
Open in a separate windowFigure 4
Two months after split‐skin transplantation. The inset photograph six weeks after transplantation. Huge amount of mesh eroded through the transplanted skin and cut off successively.
Today, our patient has begun her everyday life working 14 h per week. She has a small skin defect in the right lateral area where the native skin meets the transplanted area (Fig. (Fig.5).5). The lateral skin area especially in the right side is very heavy, and this in combination with the strong elastic belt she must use to keep the hernia in place creates friction in the wound area. Unfortunately, the patient has gained about 20 kg since discharge from the hospital, which has been counterproductive for this problem. If massive weight loss can be accomplished, reduction of the excess skin and perhaps even hernia repair could be pursued, although the latter would entail considerable risk of intestinal damage.
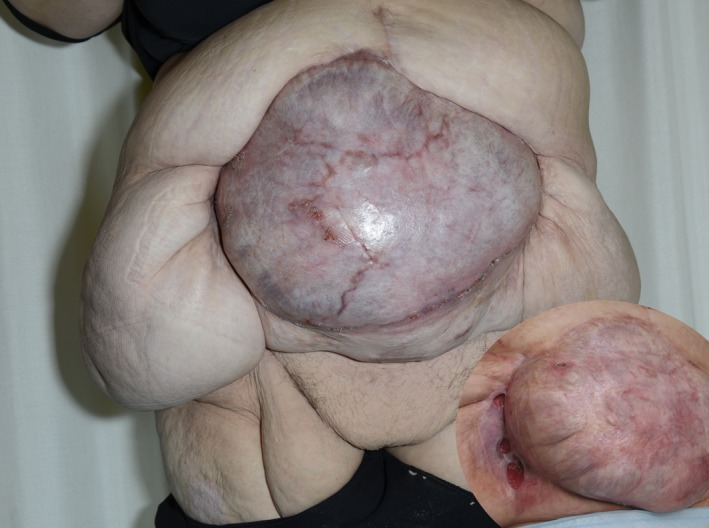
Open in a separate windowFigure 5
Nine months after skin transplantation. The patient is almost healed. Inset: 28 months after the hernia operation and 20 months after the skin transplantation. A chronic skin defect in the right lateral groove, presumably due to the heavy weight of the lateral role and mechanical collision.
References
1. Caviggioli, F. , Klinger F. M., Lisa A., Maione L., Forcellini D., Vinci V., et al. 2014. Matching biological mesh and negative pressure wound therapy in reconstructing an open abdomen defect. Case Rep. Med. 2014:235930. [PMC free article] [PubMed] [Google Scholar]2. Hutan, M. , Bartko C., Majesky I., Prochotsky A., Sekac J., and Skultety J.. 2014. Reconstruction option of abdominal wounds with large tissue defects. BMC Surg. 14:50. [PMC free article] [PubMed] [Google Scholar]3. Lee, D. L. , Ryu A. Y., and Rhee S. C.. 2011. Negative pressure wound therapy: an adjuvant to surgical reconstruction of large or difficult skin and soft tissue defects. Int. Wound J. 8:406–411. [PMC free article] [PubMed] [Google Scholar]4. Heller, L. , Chike‐Obi C., and Xue A. S.. 2012. Abdominal wall reconstruction with mesh and components separation. Semin. Plast. Surg. 26:29–35. [PMC free article] [PubMed] [Google Scholar]5. Baumann, D. P. , and Butler C. E.. 2012. Bioprosthetic mesh in abdominal wall reconstruction. Semin. Plast. Surg. 26:18–24. [PMC free article] [PubMed] [Google Scholar]6. Sacks, J. M. , Broyles J. M., and Baumann D. P.. 2012. Flap coverage of anterior abdominal wall defects. Semin. Plast. Surg. 26:36–39. [PMC free article] [PubMed] [Google Scholar]7. Fry, D. E. 2002. Wound infection in hernia repair Pp. 279–290 in Fitzgibbons R. J., Jr and Greenburg A. G., eds. Nyhus and Condon’s hernia, 5th ed. Lippincott Williams and Wilkins, Philadelphia. [Google Scholar]8. Wackenfors, A. , Sjogren J., Gustafsson R., Algotsson L., Ingemansson R., and Malmsjo M.. 2004. Effects of vacuum‐ assisted closure therapy on inguinal wound edge microvascular blood flow. Wound Repair Regen. 12:600–606. [PubMed] [Google Scholar]9. Armstrong, D. G. , Lavery L. A., and Diabetic Foot Study Consortium . 2005. Negative pressure wound therapy after partial diabetic foot amputation: a multicentre, randomised controlled trial. Lancet 366:1704–1710. [PubMed] [Google Scholar]10. Glaser, J. J. , Sheppard F. R., Gage F. A., Kumar A. R., Liston W. A., Elster E. A., et al. 2009. Warfare‐related complex abdominal wall reconstruction using a bioprosthetic regenerate template and negative pressure therapy. Eplasty 9:e17. [PMC free article] [PubMed] [Google Scholar]11. Dayton, M. T. , Buchele B. A., Shirazi S. S., Hunt L. B. 1986. Use of an absorbable mesh to repair contaminated abdominal‐wall defects. Arch. Surg. 121:954–960. [PubMed] [Google Scholar]12. Lal, P. , Kajla R. K., Chander J., Saha R., Ramteke V. K. 2003. Randomized controlled study of laparoscopic total extraperitoneal versus open Lichtenstein inguinal hernia repair. Surg. Endosc. 17:850–856. [PubMed] [Google Scholar]
Articles from Clinical Case Reports are provided here courtesy of Wiley-Blackwell
Content retrieved from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5054475/.